What are the basic properties of amino acids? What is the difference between a basic and an acidic amino acid? The basic properties are those of the side chain. In a molecule, amino acid side chains are known as peptides.
There are two forms of the peptide bond:
- trans forms
- cis forms
In addition to being optically active, amino acids have other important properties. This article will discuss the basic properties of amino acids and how these properties affect the function and structure of proteins.
Acid-base properties of amino acids

An amino acid’s acid-base properties can be found in its structure and composition. The basic properties of amino acids are essentially neutral ions, with only one -NH2 group and one -COOH group. These groups are the basis of amino acid reactions, which change the pH of a solution. An amino acid’s isoelectric point is 7 because it is neither acidic nor alkaline.
Amino acids are classified according to their structure, with one type having a carboxyl end and another with an a-carbon. These acids are also referred to as secondary amines. The difference between a-amino acids and b-amino acids lies in the side chain called R group. There are four types of amino acids: alkyl-amino acid, branched-chain peptides, and isomerized amino acids.
The acid-base properties of amino acids can be demonstrated using a potentiometric titration. In this process, the acidic form of alanine is titrated by adding NaOH to a solution. A graph is then plotted that shows the change in pH as the amino acid molecules are added. Both the pH and the volume of the titrant are recorded. In this way, one can determine the pH of a solution without having to worry about determining the concentration of an acid.
Despite the fact that these compounds are neutral, there are still many misconceptions about their acids. While they are used in everyday life, they also have industrial applications. Their chemical properties and applications range from the production of drugs to biodegradable plastics. One way to understand their properties is to try the reactions they are involved in with food and beverages. Aside from that, amino acids are often used in the production of biodegradable plastics and chiral catalysts.
Colorless
The simplest basic properties of amino acid is glycine, which is a sweet-tasting, colorless crystalline solid. It is nonvolatile and melts at temperatures above 200degC. These temperatures are higher than those of organic acids and amines. The chemical structure of an amino acid consists of one positively and one negatively charged group, known as a zwitterion. It is therefore considered the simplest type of amino acid.
The chirality of an amino acid is determined by its R-substituent, which is a three-carbon chain attached to an alpha-carbon ring. The chiral nature of amino acids is one of the main factors determining their colorlessness and their structural differences. Cys is the only natural colorless amino acid. The chiral structure of the acid is based on Cahn-Ingold-Prelog priority rules, which are widely used in chemistry.
Crystalline

Amino acids are crystalline solids that melt at temperatures of 200degC or higher. They are more like inorganic salts than organic acids or amines because of the positive and negative charge on the amine group. Crystalline ammonium acetate has a zwitterion structure. As a result, the amino acid has a higher melting point than other organic acids. In addition, amino acids are insoluble in non-polar organic solvents.
In addition to their molecular mass, amino acids are chiral. They do not have a plane of symmetry and have two stereoisomers. This gives rise to their crystalline nature. This is also true of their structure. They have been investigated by spectroscopy and piezoelectric response at temperatures from 120 K to 320 K. And finally, they were studied by optical second harmonic generation and optical activity.
Each amino acid has a different structure. It is distinguished by its amine and acid functional groups. Despite these differences, each amino acid has special common names, such as a-amino-proline. The abbreviations for residues with multiple identities are included in blue. The termination codon is shown in red. This structure can be useful for identifying the type of amino acid required for a particular species.
Aside from being the building blocks of proteins, amino acids also determine a wide range of biochemical properties. The amino acid cysteine, for example, can be converted into a crystalloid form by replacing a portion of soybean meal with a crystalline form. This synthetic form is 100 percent digested and decomposed by bacteria. This means that crystalline amino acids are ideal for the production of proteins since they are non-toxic to humans and the environment.
Physical properties of amino acids
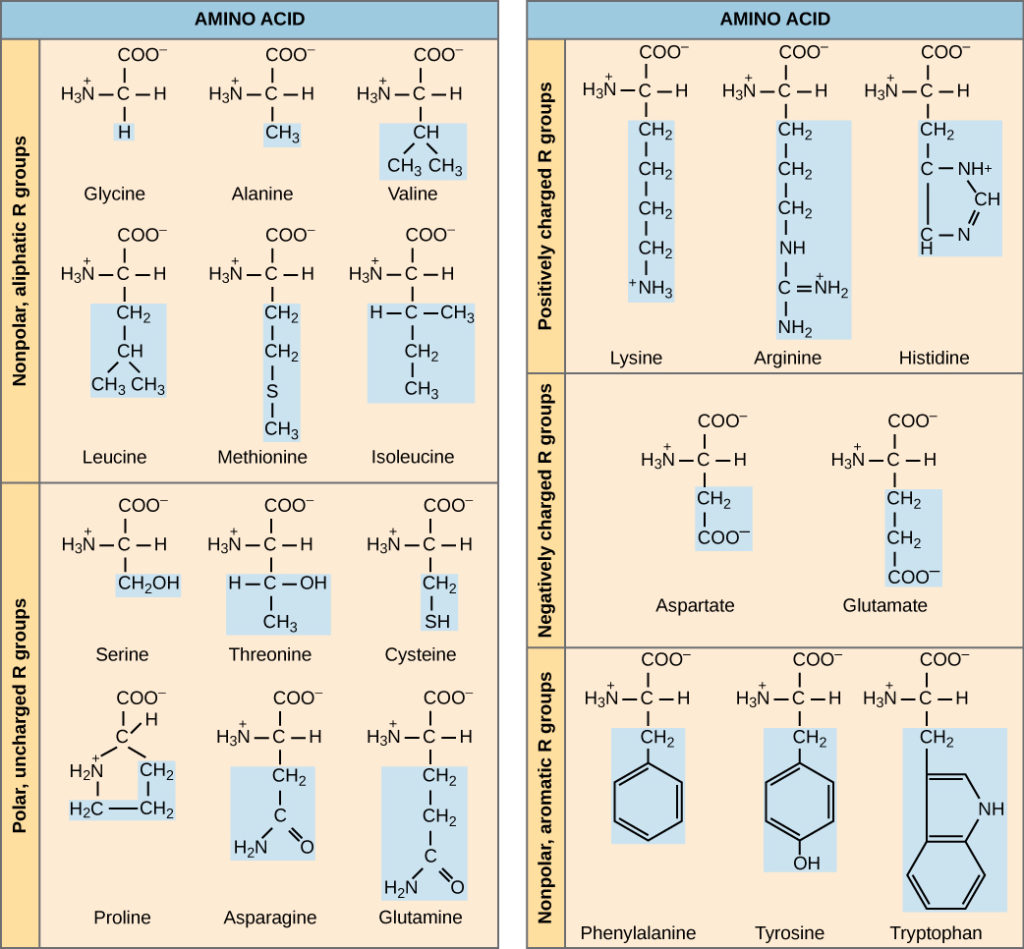
There are two main types of amino acids: polar and nonpolar. The polar amino acids are those that contain a hydrocarbon side chain, and the nonpolar ones are those that lack this ring structure. Nonpolar amino acids include asparagine, cysteine, leucine, and phenylalanine, as well as any amino acid with an aromatic ring. Depending on the amino acid, nonpolar or polar amino acids may be more or less acidic.
Each amino acid consists of five different atoms – H, C, N, and O. The polarity of an amino acid is determined by its alkyl groups, with the higher the number, the less polar it is. Valine and methionine are two examples of nonpolar amino acids, which are water-fearing. However, polar amino acids are not inherently anti-magnetic.
Amino acids are classified based on their location in protein molecules, as well as their degree of exposure to solvent. Amino acids with hydrophobic side chains are usually buried, while those with polar or charged residues are exposed to greater extents. In addition, polar amino acids have higher pKa values than nonpolar acids. However, the chemical reactions involving amino acids require the extraction of a proton.
Glycine is a common amino acid that is found in coils and loops. This amino acid gives a polypeptide chain its high flexibility, which is important for sharp turns in loop regions. On the other hand, proline is a nonpolar amino acid that imparts rigidity to a polypeptide chain. Its side chain forms a covalent bond with the main chain and constrains the polypeptide’s phi-angle.
Hydrophobic
Quantitative characterization of the hydrophobic basic properties of amino acids is an important prerequisite for predicting the structures and functionalities of proteins in a biological environment. These properties of amino acids can be measured using phase-partitioning behavior of molecular fragments. The corresponding values for hydrophobic amino acid fractions can be obtained by calculating the partitioning coefficients of the aliphatic and phenolic hydroxyls of the amino acids.
The hydrophobic basic properties of amino acids are associated with their lack of polarity in their side chains. These amino acids generally reside in the hydrophobic core of proteins and lipid portions of membranes. These properties are describe by authors including M.J. Betts, R.B. Russell, and M.R. Barnes. I.C. Gray is the editor of Bioinformatics for Geneticists.
The hydrophobicity of a macroscopic planar surface is typically measured by the contact angle of water droplets, but this cannot be extended to proteins due to their nanometer-scale amino acids and inherent nonplanar structure. Hydrophobic solvation is characterized by numerous parameters. However, a consistent pattern cannot be established for all parameters. Hydrophobic basic properties of amino acids can be characterized using computational data.
A combination of Pronase and Peptidase R shows the highest selectivity for hydrophobic amino acids. The enzymes increased hydrophobic amino acid concentrations by twofold and doubled their selectivity. This enzyme-based method is applicable to agro-industrial residues. It have been shown that Pronase and Peptidase R can increase the hydrophobic basic content of proteins by combining them.



